Topshots of the Week (10-16 February 2024)
A selection of some of the most striking photographs taken by AFP photojournalists from the 10th to the 16th of February 2024.

Taipei (AFP) | 27/05/2026 - 06:37:55 | Nvidia to boost spending in Taiwan to $150 bn a year: CEO
Rio de Janeiro (AFP) | 27/05/2026 - 05:01:14 | Deforestation in Brazil's Amazon falls to six-year low: report
Seoul (AFP) | 27/05/2026 - 03:33:28 | Samsung labour union passes bonus deal: union
La Paz (AFP) | 27/05/2026 - 03:17:07 | Bolivian Congress OK's use of troops against protesters
Seoul (AFP) | 27/05/2026 - 00:07:02 | N. Korea tests new multi-purpose missile launch system: KCNA
Washington (AFP) | 27/05/2026 - 00:03:43 | IMF says Bangladesh requests new assistance program amid Iran war fallout
Beirut (AFP) | 26/05/2026 - 23:07:26 | Lebanon says Israeli strikes kill 31 on Tuesday
Jerusalem (AFP) | 26/05/2026 - 20:54:06 | Israeli military issues evacuation warning for 19 south Lebanon villages
Jerusalem (AFP) | 26/05/2026 - 20:22:15 | Israel says targeted new Hamas armed wing chief in Gaza strike
Washington (AFP) | 26/05/2026 - 18:51:58 | Trump says medical check-up went 'perfectly'
A selection of some of the most striking photographs taken by AFP photojournalists from the 10th to the 16th of February 2024.

A selection of some of the most striking photographs taken by AFP photojournalists from the 3rd of February to the 9th of February 2024.

A selection of some of the most striking photographs taken by AFP photojournalists from the 27th of January to the 2nd of February 2024.
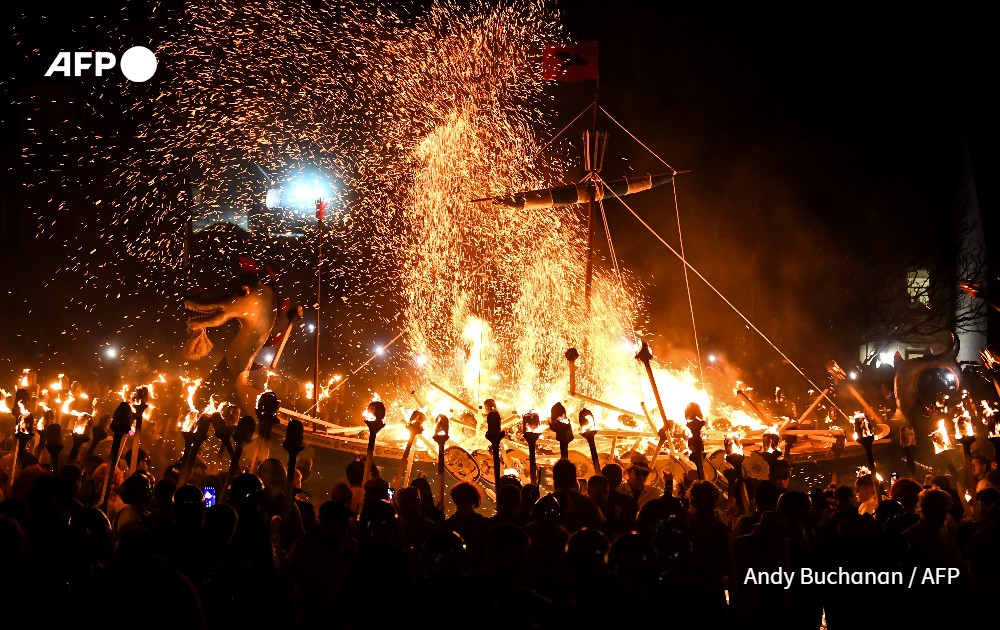
A selection of some of the most striking photographs taken by AFP photojournalists from the 20th to the 26th of January 2024.

A selection of some of the most striking photographs taken by AFP photojournalists from the 13th of January to the 19th of January 2024.
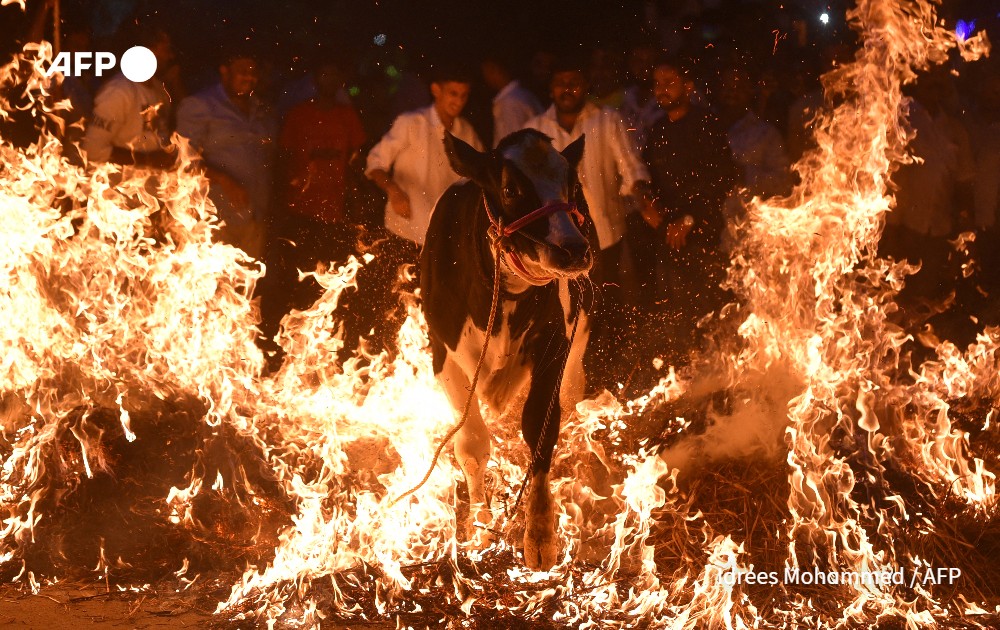
Welcome back to Your Planet, a podcast series brought to you in association with The Conversation that delves into potential solutions for addressing climate change and the ecological crisis.

A selection of some of the most striking photographs taken by AFP photojournalists from the 6th of January to the 12th of January 2024.

A selection of some of the most striking photographs taken by AFP photojournalists from the 30th of December 2023 to the 5th of January 2024.

Fireworks explode over the Sydney Harbour Bridge and Sydney Opera House during New Year's Eve celebrations in Sydney on January 1, 2024.
© Izhar KHAN / AFP
Welcome back to Your Planet, a podcast series brought to you in association with The Conversation that delves into potential solutions for addressing climate change and the ecological crisis.

A selection of some of the most striking photographs taken by AFP photojournalists from the 23rd of December to the 29th of December 2023.
